
Hyperphosphatemia: Practice Essentials, Background, Pathophysiology. Hyperphosphatemia, that is, an abnormally high serum phosphate level, can result from increased phosphate (PO4) intake, decreased phosphate excretion, or a disorder that shifts intracellular phosphate to extracellular space. A highly reactive substance, it occurs in nature, including in the human body, as phosphate. Phosphorus (phosphate) is critical for bone mineralization, cellular structure, genetic coding, and energy metabolism. The adult body contains approximately 1. An additional 1. 0- 1.


Phosphorus is present in nearly all foods, and gastrointestinal (GI) absorption of dietary forms is very efficient. With low dietary intake, 8. When intake is greater than 1. Normal daily dietary intake varies from 8.
Absorption occurs mainly in the jejunum, although some absorption occurs throughout the GI tract. A small amount of phosphorus is secreted into the GI tract. Free serum compounds represent much less than 1% of the total body phosphorus content.
Q.1- A 64 year-old man who develops acute renal failure while recovering from a severe acute myocardial infarction (Acute MI), Blood chemistry reveals.
This fraction also varies with shifts between the intracellular and extracellular compartments. Thus, serum phosphorus levels may not reflect accurately the total body phosphorus content.
One mg/d. L of phosphorus is equal to 0. The normal adult range for phosphorus is 2. L (0. 8. 1- 1. 4.
L). Levels are 5. Hyperphosphatemia is considered significant when levels are greater than 5 mg/d. L in adults or 7 mg/d. L in children or adolescents. In addition to providing mineral strength to bone, it is an integral component of the nucleic acids that make up deoxyribonucleic acid (DNA) and ribonucleic acid (RNA). The phosphate bonds of adenosine triphosphate (ATP) carry the energy required for all cellular functions.
International programme on chemical safety world health organization toxicological evaluation of certain veterinary drug residues in food who food additives. ARUP Laboratories is a national reference laboratory and a worldwide leader in innovative laboratory research and development. ARUP offers an extensive lab testing.
Online Medical Dictionary and glossary with medical definitions, p listing. Emeritus Professor of Medicine, Physiology and Biophysics School of Medicine. Several forms of vitamin D exist. The two major forms are vitamin D 2 or ergocalciferol, and vitamin D 3 or cholecalciferol; vitamin D without a subscript refers to. A Trial of Darbepoetin Alfa in Type 2 Diabetes and Chronic Kidney Disease. Pfeffer, M.D., Ph.D., Emmanuel A. Burdmann, M.D., Ph.D., Chao-Yin. Columbus Instruments' Comprehensive Lab Animal Monitoring System (CLAMS) has set the standard for multiple parameter scoring of multiple animals.
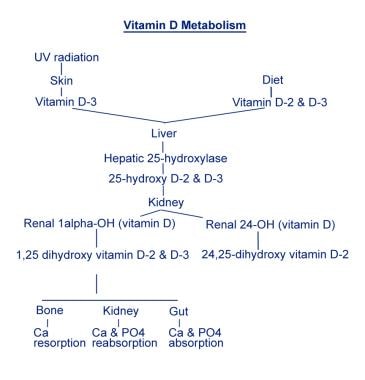
The addition and deletion of phosphate groups to enzymes and proteins are common mechanisms for the regulation of their activity. Phosphate also functions as a buffer in bone, serum, and urine. In view of the sheer breadth of influence of phosphorus, phosphate homeostasis (as depicted in the image below) is understandably a highly regulated process. As previously stated, 8. This phosphate pool is accessible, albeit in a somewhat limited fashion. Approximately 3. 00 mg of phosphate enters and exits bone tissue each day. Excessive losses or failure to add phosphate to bone leads to osteomalacia.
Intracellular concentration and transport. Phosphate is a predominantly intracellular anion (it has a negative charge) with a concentration of approximately 1. L, although determination of the precise intracellular concentration has been difficult. Most intracellular phosphate is either complexed or bound to proteins or lipids. In response to kinases and phosphatases, these phosphate ions attach and detach from different molecules, forming a constantly shifting pool.
Intracellular phosphate is essential for most, if not all, cellular processes; however, because the intracellular concentration of phosphate is greater than the extracellular concentration, phosphate entry into cells requires a facilitated transport process. Several sodium- coupled transport proteins have been identified that enable intracellular uptake of phosphate by taking advantage of the steep extracellular- to- intracellular sodium gradient. Type 1 sodium phosphate cotransporters are expressed predominantly in kidney cells on the apical membranes of proximal tubule cells and liver. They are capable of transporting organic ions and stimulating chloride conductance in addition to phosphate. Their role in phosphate homeostasis is not clear.
Other sites of expression include the liver and brain. Type 2 sodium phosphate cotransporters are expressed in kidneys, bone, intestines, and a variety of other epithelial tissues responsible for transepithelial transport. Type 2a transporters are expressed in the apical membranes of kidney proximal tubules, are very specific for phosphate, and are regulated by several physiologic mediators of phosphate homeostasis, such as parathyroid hormone (PTH), dopamine, and dietary phosphate. Currently, these transporters are believed to be most critical for maintenance of renal phosphate homeostasis.
Impaired expression or function of these transporters is associated with nephrolithiasis. They are expressed in the small intestine and are also upregulated under conditions of dietary phosphate deprivation. Many additional epithelial tissues express type 2b transporters, such as mammary glands, lung epithelium, salivary glands, and testis. It is unlikely that the transporters at these sites contribute to phosphate homeostasis; however, the absence of this transporter can be associated with clinical conditions such as broncholithiasis.
Type 2c transporters, a third member of the Type 2 sodium phosphate cotransporter family, were initially described as growth- related phosphate transporters. They are expressed exclusively on the S1 segment of the proximal tubule and together with Type 2a transporters are essential for normal phosphate homeostasis. Similarly to type 2a transporters, type 2c transporters are also regulated by diet and PTH. Loss of type 2c function results in hereditary hypophosphatemic rickets with hypercalciuria. Almost all cells express type 3 sodium phosphate cotransporters; therefore, these transporters presumably play a housekeeping role in ensuring adequate phosphate for all cells. The factors that regulate the activity of these transporter proteins are not completely understood.
Evidence suggests, however, that these transporters may also participate in regulation of renal and intestinal transepithelial transport . Because the ionization constant of acid (p. K) of phosphate is 6. H of 7. 4 the univalent species is 4 times as prevalent as the divalent species.
Serum phosphate concentration varies with age, time of day, fasting state, and season. Serum phosphate concentration is higher in children than adults; the reference range is 4- 7 mg/d. L in children compared with 2. L in adults. A diurnal variation exists, with the highest phosphate level occurring near noon.
Serum phosphate concentration is regulated by diet, hormones, and physical factors such as p. H. Importantly, because phosphate moves in and out of cells under several influences, the serum concentration of phosphate may not reflect true phosphate stores. Often, persons with alcoholism who have severely deficient phosphate stores may present for medical treatment with a normal serum phosphate level. Only after refeeding will serum phosphate levels decline, often abruptly plummeting to dangerously low levels. Phosphate homeostasis. Phosphate is plentiful in the diet. A normal diet provides approximately 1.
The fractional absorption of phosphate can be increased by increasing vitamin D intake and by ingesting a very low. Under these conditions, the intestine expresses sodium- coupled phosphate transporters to enhance phosphate uptake. Regulation of intestinal phosphate transport overall is poorly understood. Although studies had suggested that the majority of small intestine phosphate uptake was accomplished through unregulated, sodium- independent pathways, subsequent investigations have suggested that regulated sodium- dependent mechanisms may play a greater role in overall intestinal phosphate handling than was previously appreciated. Furthermore, intestinal cells may have a role in renal phosphate handling through elaboration of circulating phosphaturic substances in response to sensing a phosphate load. Mild to moderate use of such phosphate binders generally poses no threat to phosphate homeostasis, because dietary ingestion greatly exceeds body needs.
However, very heavy use of these antacids can cause significant phosphate deficits. Stool losses of phosphate are minor; ie, 1. However, diseases that cause severe diarrhea or intestinal malabsorption can dramatically increase these losses. Bone loses approximately 3.
Bone metabolism of phosphate is influenced by factors that determine bone formation and destruction; ie, PTH, vitamin D, sex hormones, acid- base balance, and inflammatory status. PTH and vitamin DExcess ingested phosphate is excreted by the kidneys to maintain phosphate balance. Major sites of regulation of phosphate excretion are the early proximal renal tubule and the distal convoluted tubule. In the proximal tubule, phosphate reabsorption by type 2 sodium phosphate cotransporters is regulated by dietary phosphate, PTH, and vitamin D. High dietary phosphate intake and elevated PTH levels decrease proximal renal tubule phosphate absorption, thus enhancing renal excretion. Defense against hyperphosphatemia is depicted in the image below. Hyperphosphatemia inhibits 1- alpha hydroxylase in the proximal tubule directly and indirectly through stimulation of FGF2.
D3 to the active metabolite, 1,2. D3. FGF2. 3 additionally increases the expression of 2. D3. The decrease in active vitamin D production with high phosphate is somewhat offset by the ability of hyperphosphatemia to stimulate the secretion of parathyroid hormone (PTH), which will increase the activity of 1- alpha hydroxylase.
The result is generally a neutral effect on intestinal phosphate absorption. Hyperphosphatemia- stimulated PTH secretion is mediated through an as yet unidentified pathway. With normal renal function, the transient increase in PTH and decrease in vitamin D serve to inhibit renal and intestinal absorption of phosphate, resulting in resolution of the hyperphosphatemia. In contrast, under conditions of renal failure, sustained hyperphosphatemia results in sustained hyperparathyroidism. The hyperparathyroidism enhances renal phosphate excretion but also enhances bone resorption, releasing more phosphate into the serum.
As renal failure progresses and the ability of the kidney to excrete phosphate continues to diminish, the action of PTH on the bone can exacerbate the already present hyperphosphatemia. Conversely, low dietary phosphate intake, low PTH levels, and high vitamin D levels enhance renal proximal tubule phosphate absorption.
Vitamin D - Wikipedia. Vitamin D refers to a group of fat- soluble secosteroids responsible for increasing intestinal absorption of calcium, iron, magnesium, phosphate, and zinc. In humans, the most important compounds in this group are vitamin D3 (also known as cholecalciferol) and vitamin D2 (ergocalciferol). Vitamin D is made in the skin from cholesterol dependent on sun exposure (specifically UVB radiation). Vitamin D from the diet or dermal synthesis from sunlight is biologically inactive; activation requires enzymatic conversion (hydroxylation) in the liver and kidney. Evidence indicates the synthesis of vitamin D from sun exposure is regulated by a negative feedback loop that prevents toxicity, but because of uncertainty about the cancer risk from sunlight, no recommendations are issued by the Institute of Medicine (US) for the amount of sun exposure required to reach vitamin D requirements.
Accordingly, the Dietary Reference Intake for vitamin D assumes no synthesis occurs and all of a person's vitamin D is from food intake. As vitamin D is synthesized in adequate amounts by most mammals exposed to sunlight. Its discovery was due to effort to find the dietary substance lacking in rickets (the childhood form of osteomalacia).
Ergocalciferol (vitamin D2) is converted in the liver to 2. D2 . These two specific vitamin D metabolites are measured in serum to determine a person's vitamin D status. Calcitriol also affects neuromuscular and immune function. The two major forms are vitamin D2 or ergocalciferol, and vitamin D3 or cholecalciferol; vitamin D without a subscript refers to either D2 or D3 or both. These are known collectively as calciferol. In 1. 93. 5, the chemical structure of vitamin D3 was established and proven to result from the ultraviolet irradiation of 7- dehydrocholesterol.
In the developed world, this is a rare disease. This condition is characterized by bow legs. In sunny countries such as Nigeria, South Africa, and Bangladesh, where the disease occurs among older toddlers and children, it has been attributed to low dietary calcium intakes, which are characteristic of cereal- based diets with limited access to dairy products.
Characteristics of this disease are softening of the bones, leading to bending of the spine, bowing of the legs, proximal muscle weakness, bone fragility, and increased risk for fractures. Osteomalacia is usually present when 2. D levels are less than about 1. L. Black women have an increase in serum parathyroid hormone at a lower 2. OH)D level than white women. The threshold for vitamin D toxicity has not been established; however, according to some research, the tolerable upper intake level (UL) is 4,0.
IU/day for ages 9. The FDA advised manufacturers of liquid vitamin D supplements that droppers accompanying these products should be clearly and accurately marked for 4. IU). In addition, for products intended for infants, the FDA recommends the dropper hold no more than 4. IU. One thousand micrograms per day in infants has produced toxicity within one month. If hypercalcemia is not treated, it results in excess deposits of calcium in soft tissues and organs such as the kidneys, liver, and heart, resulting in pain and organ damage. Kidney damage may be irreversible.
Exposure to sunlight for extended periods of time does not normally cause vitamin D toxicity. The concentrations of vitamin D precursors produced in the skin reach an equilibrium, and any further vitamin D produced is degraded. The review concluded that ranges from 7. L were to be recommended for athletes.
Other countries often use nmol/l; ng x 2. In 2. 01. 1 an IOM committee concluded a serum 2. D level of 2. 0 ng/ml (5. The dietary reference intakes for vitamin D are chosen with a margin of safety and 'overshoot' the targeted serum value to ensure the specified levels of intake achieve the desired serum 2.
D levels in almost all persons. No contributions to serum 2.
D level are assumed from sun exposure and the recommendations are fully applicable to people with dark skin or negligible exposure to sunlight. Serum 2. 5- hydroxyvitamin D levels above 5. Research on vitamin D supplements, including large- scale clinical trials, is continuing. Use of vitamin D in children with normal vitamin D levels does not appear to improve bone density.
As of August 2. 01. The greatest benefit with supplementation is seen in athletes who are deficient (2. OH)D serum levels < 3. OH)D serum levels < 2. Incremental decreases in risks are observed with rising serum 2. OH)D concentrations plateauing at 5.
Reviews have described the evidence as being . However, lower vitamin D concentrations is also associated with poor nutrition and spending less time outdoors. Therefore, alternative explanations for the increase in cognitive impairment exist and hence a direct causal relationship between vitamin D levels and cognition could not be established. Circulating calcifediol may then be converted into calcitriol, the biologically active form of vitamin D, in the kidneys. Following the final converting step in the kidney, calcitriol is released into the circulation.
By binding to vitamin D- binding protein, a carrier protein in the plasma, calcitriol is transported to various target organs. When synthesized by monocyte- macrophages, calcitriol acts locally as a cytokine, defending the body against microbial invaders by stimulating the innate immune system.
This reaction is catalyzed by the microsomal enzyme vitamin D 2. Once made, the product is released into the plasma, where it is bound to an .
This product is a potent ligand of the vitamin D receptor, which mediates most of the physiological actions of the vitamin. The conversion of calcifediol to calcitriol is catalyzed by the enzyme 2. D3 1- alpha- hydroxylase, the levels of which are increased by parathyroid hormone (and additionally by low calcium or phosphate). Biosynthesis. Second, previtamin D3 spontaneously isomerizes to vitamin D3 (cholecalciferol) in an antarafacialsigmatropic .
At room temperature, the transformation of previtamin D3 to vitamin D3 in an organic solvent takes about 1. The conversion of previtamin D3 to vitamin D3 in the skin is about 1. Although primitive vertebrates in the ocean could absorb calcium from the ocean into their skeletons and eat plankton rich in vitamin D, land animals required another way to satisfy their vitamin D requirement for a calcified skeleton without relying on plants. Land vertebrates have been making their own vitamin D for more than 3. Dehydrocholesterol reacts with UVB light at wavelengths between 2.
Exposure to light through windows is insufficient because glass almost completely blocks UVB light. The darker the skin, and the weaker the sunlight, the more minutes of exposure are needed. Vitamin D overdose is impossible from UV exposure; the skin reaches an equilibrium where the vitamin degrades as fast as it is created. Vitamin D is produced in the keratinocytes. In birds and fur- bearing mammals, vitamin D is generated from the oily secretions of the skin deposited onto the feathers or fur and is obtained orally during grooming.
VDR activation in the intestine, bone, kidney, and parathyroid gland cells leads to the maintenance of calcium and phosphorus levels in the blood (with the assistance of parathyroid hormone and calcitonin) and to the maintenance of bone content. Vitamin D deficiency can result in lower bone mineral density and an increased risk of reduced bone density (osteoporosis) or bone fracture because a lack of vitamin D alters mineral metabolism in the body. Vitamin D also affects the immune system, and VDRs are expressed in several white blood cells, including monocytes and activated T and B cells.
British doctor Edward Mellanby noticed dogs that were fed cod liver oil did not develop rickets and concluded vitamin A, or a closely associated factor, could prevent the disease. In 1. 92. 2, Elmer Mc. Collum tested modified cod liver oil in which the vitamin A had been destroyed. He called it vitamin D because it was the fourth vitamin to be named. Alfred Fabian Hess stated: . A meeting took place with J.
B. S. Bernal, and Dorothy Crowfoot to discuss possible structures, which contributed to bringing a team together. X- ray crystallography demonstrated the sterol molecules were flat, not as proposed by the German team led by Windaus. In 1. 93. 2, Otto Rosenheim and Harold King published a paper putting forward structures for sterols and bile acids which found immediate acceptance. In the 1. 93. 0s, Windaus clarified further the chemical structure of vitamin D. A vitamin D deficiency is a known cause of rickets.
Using $3. 00 of his own money, Steenbock patented his invention. His irradiation technique was used for foodstuffs, most memorably for milk. By the expiration of his patent in 1. US.<. In the liver, vitamin D was found to be converted to calcifediol. Calcifediol is then converted by the kidneys to calcitriol, the biologically active form of vitamin D. The vitamin D metabolites, calcifediol and calcitriol, were identified by competing teams led by Michael F. Holick in the laboratory of Hector De.
Luca and by Tony Norman and colleagues. The recommended daily intake of vitamin D may not be sufficient if sunlight exposure is limited. This dose should be increased to 4,0. IU/day in some patients with very low vitamin D status or in case of comorbid conditions. For vitamin D labeling purposes 1.
Daily Value was 4. IU (1. 0 . A table of the pre- change adult Daily Values is provided at Reference Daily Intake. Food and supplement companies have until July 2. Upper intake levels. The recommendations were formed assuming the individual has no skin synthesis of vitamin D because of inadequate sun exposure. The reference intake for vitamin D refers to total intake from food, beverages and supplements, is intended for the North American population, and assumes that calcium requirements are being met. Proponents of this view, who include some members of the panel that drafted a now- superseded 1.
Risperidone Full Prescribing Information - Psychiatric Medications - Other Info. Risperdal is an atypical antipsychotic used for the treatment of schizophrenia in adults; and the treatment of bipolar mania associated with Bipolar I Disorder in adults and children (ages 1. Uses, dosage, side effects of Risperdal below.
Contents: Description. Pharmacology. Indications and Usage. Contraindications. Warnings. Precautions. Drug Interactions.
Adverse Reactions. Overdose. Dosage. Supplied. Risperdal patient information (in plain English)Increased Mortality in Elderly Patients with Dementia - Related Psychosis Elderly patients with dementia- related psychosis treated with atypical antipsychotic drugs are at an increased risk of death compared to placebo. Analyses of seventeen placebo controlled trials (modal duration of 1. Over the course of a typical 1. Although the causes of death were varied, most of the deaths appeared to be either cardiovascular (e.
Its molecular formula is C2. H2. 7FN4. O2 and its molecular weight is 4. The structural formula is: Risperidone is a white to slightly beige powder.
It is practically insoluble in water, freely soluble in methylene chloride, and soluble in methanol and 0. N HCl. RISPERDALtablets are available in 0. Inactive ingredients are colloidal silicon dioxide, hypromellose, lactose, magnesium stearate, microcrystalline cellulose, propylene glycol, sodium lauryl sulfate, and starch (corn).
Tablets of 0. 2. 5, 0. The 0. 2. 5 mg tablets contain yellow iron oxide; the 0. FD& C Yellow No. Aluminum Lake; the 3 mg and 4 mg tablets contain D& C Yellow No. FD& C Blue No. Aluminum Lake. RISPERDALis also available as a 1 mg/m.
L oral solution. The inactive ingredients for this solution are tartaric acid, benzoic acid, sodium hydroxide, and purified water. RISPERDALM- TABOrally Disintegrating Tablets are available in 0. RISPERDALM- TABOrally Disintegrating Tablets contain the following inactive ingredients: Amberlite resin, gelatin, mannitol, glycine, simethicone, carbomer, sodium hydroxide, aspartame, red ferric oxide, and peppermint oil. Clinical Pharmacology. Pharmacodynamics.
The mechanism of action of RISPERDAL. However, it has been proposed that the drug's therapeutic activity in schizophrenia is mediated through a combination of dopamine Type 2 (D2) and serotonin Type 2 (5. HT2) receptor antagonism. Antagonism at receptors other than D2 and 5.
HT2 may explain some of the other effects of RISPERDAL. The absolute oral bioavailability of risperidone is 7. CV=2. 5%). The relative oral bioavailability of risperidone from a tablet is 9. CV=1. 0%) when compared to a solution.
Pharmacokinetic studies showed that RISPERDAL. Following oral administration of solution or tablet, mean peak plasma concentrations of risperidone occurred at about 1 hour. Peak concentrations of 9- hydroxyrisperidone occurred at about 3 hours in extensive metabolizers, and 1. Steady- state concentrations of risperidone are reached in 1 day in extensive metabolizers and would be expected to reach steady- state in about 5 days in poor metabolizers. Steady- state concentrations of 9- hydroxyrisperidone are reached in 5- 6 days (measured in extensive metabolizers). Food Effect Food does not affect either the rate or extent of absorption of risperidone. Thus, risperidone can be given with or without meals.
Distribution. Risperidone is rapidly distributed. The volume of distribution is 1- 2 L/kg. In plasma, risperidone is bound to albumin and a- acid glycoprotein.
The plasma protein binding of risperidone is 9. Neither risperidone nor 9- hydroxyrisperidone displaces each other from plasma binding sites. High therapeutic concentrations of sulfamethazine (1. L), warfarin (1. 0 mcg/m.
L), and carbamazepine (1. L) caused only a slight increase in the free fraction of risperidone at 1. L and 9- hydroxyrisperidone at 5. L, changes of unknown clinical significance. Metabolism. Risperidone is extensively metabolized in the liver. The main metabolic pathway is through hydroxylation of risperidone to 9- hydroxyrisperidone by the enzyme, CYP 2. D6. A minor metabolic pathway is through N- dealkylation.
The main metabolite, 9- hydroxyrisperidone, has similar pharmacological activity as risperidone. Consequently, the clinical effect of the drug (e. CYP 2. D6, also called debrisoquin hydroxylase, is the enzyme responsible for metabolism of many neuroleptics, antidepressants, antiarrhythmics, and other drugs. CYP 2. D6 is subject to genetic polymorphism (about 6%- 8% of Caucasians, and a very low percentage of Asians, have little or no activity and are . Extensive CYP 2. D6 metabolizers convert risperidone rapidly into 9- hydroxyrisperidone, whereas poor CYP 2.
D6 metabolizers convert it much more slowly. Although extensive metabolizers have lower risperidone and higher 9- hydroxyrisperidone concentrations than poor metabolizers, the pharmacokinetics of the active moiety, after single and multiple doses, are similar in extensive and poor metabolizers. Risperidone could be subject to two kinds of drug- drug interactions (see PRECAUTIONS - Drug Interactions). First, inhibitors of CYP 2. D6 interfere with conversion of risperidone to 9- hydroxyrisperidone. This occurs with quinidine, giving essentially all recipients a risperidone pharmacokinetic profile typical of poor metabolizers. The therapeutic benefits and adverse effects of risperidone in patients receiving quinidine have not been evaluated, but observations in a modest number (n 7.
Second, co- administration of known enzyme inducers (e. It would also be possible for risperidone to interfere with metabolism of other drugs metabolized by CYP 2. D6. Relatively weak binding of risperidone to the enzyme suggests this is unlikely. In a drug interaction study in schizophrenic patients, 1.
During co- administration, the plasma concentrations of risperidone and its pharmacologically active metabolite, 9- hydroxyrisperidone, were decreased by about 5. Plasma concentrations of carbamazepine did not appear to be affected. Co- administration of other known enzyme inducers (e.
PRECAUTIONS - Drug Interactions and DOSAGE AND ADMINISTRATION - Co- Administration of RISPERDAL. Fluoxetine did not affect the plasma concentration of 9- hydroxyrisperidone. Paroxetine lowered the concentration of 9- hydroxyrisperidone an average of 1. PRECAUTIONS - Drug Interactions and DOSAGE AND ADMINISTRATION - Co- Administration of RISPERDAL.
However, there was a 2. Cmax) after concomitant administration of risperidone (see PRECAUTIONS - Drug Interactions).
There were no significant interactions between risperidone (1 mg QD) and erythromycin (5. QID) (see PRECAUTIONS - Drug Interactions). Excretion. Risperidone and its metabolites are eliminated via the urine and, to a much lesser extent, via the feces. As illustrated by a mass balance study of a single 1 mg oral dose of C- risperidone administered as solution to three healthy male volunteers, total recovery of radioactivity at 1 week was 8. The apparent half- life of risperidone was 3 hours (CV=3. CV=4. 0%) in poor metabolizers.
The apparent half- life of 9 - hydroxyrisperidone was about 2. CV=2. 0%) in extensive metabolizers and 3. CV=2. 5%) in poor metabolizers.
The pharmacokinetics of the active moiety, after single and multiple doses, were similar in extensive and poor metabolizers, with an overall mean elimination half- life of about 2. Special Populations Renal Impairment In patients with moderate to severe renal disease, clearance of the sum of risperidone andits active metabolite decreased by 6. Dosing should be modified accordingly in the elderly patients (see DOSAGE AND ADMINISTRATION). Race and Gender Effects No specific pharmacokinetic study was conducted to investigate race and gender effects, but a population pharmacokinetic analysis did not identify important differences in the disposition of risperidone due to gender (whether corrected for body weight ornot) or race.
Clinical Trials Schizophrenia. Short- Term Efficacy The efficacy of RISPERDAL. The BPRS psychosis cluster (conceptual disorganization, hallucinatory behavior, suspiciousness, and unusual thought content) is considered a particularly useful subset for assessing actively psychotic schizophrenic patients. A second traditional assessment, the Clinical Global Impression (CGI), reflects the impression of a skilled observer, fully familiar with the manifestations of schizophrenia, about the overall clinical state of the patient.
In addition, the Positive and Negative Syndrome Scale (PANSS) and the Scale for Assessing Negative Symptoms (SANS) were employed. The results of the trials follow: (1) In a 6- week, placebo- controlled trial (n=1. RISPERDAL. The most consistently positive responses on all measures were seen for the 6 mg dose group, and there was no suggestion of increased benefit from larger doses.(3) In an 8- week, dose comparison trial (n=1. RISPERDAL. None of the dose groups were superior to the 1 mg group on the PANSS negative subscale. The most consistently positive responses were seen for the 4 mg dose group.
In a 4- week, placebo- controlled dose comparison trial (n=2. RISPERDAL. The results were generally stronger for the 8 mg than for the 4 mg dose group.
Long- Term Efficacy In a longer- term trial, 3. DSM- IV criteria for schizophrenia and who had been clinically stable for at least 4 weeks on an antipsychotic medication were randomized to RISPERDAL. Patients receiving RISPERDAL. These trials included patients with or without psychotic features. The primary rating instrument used for assessing manic symptoms in these trials was the Young Mania Rating Scale (Y- MRS), an 1. The primary outcome in these trials was change from baseline in the Y- MRS total score.
The results of the trials follow: (1) In one 3- week placebo- controlled trial (n=2. RISPERDAL. This trial included patients with or without psychotic features and with or without a rapid- cycling course.(1) In this 3- week placebo- controlled combination trial, 1.
 RSS Feed
RSS Feed
